Unknown number 1, which patients with good prognosis melanoma still relapse?
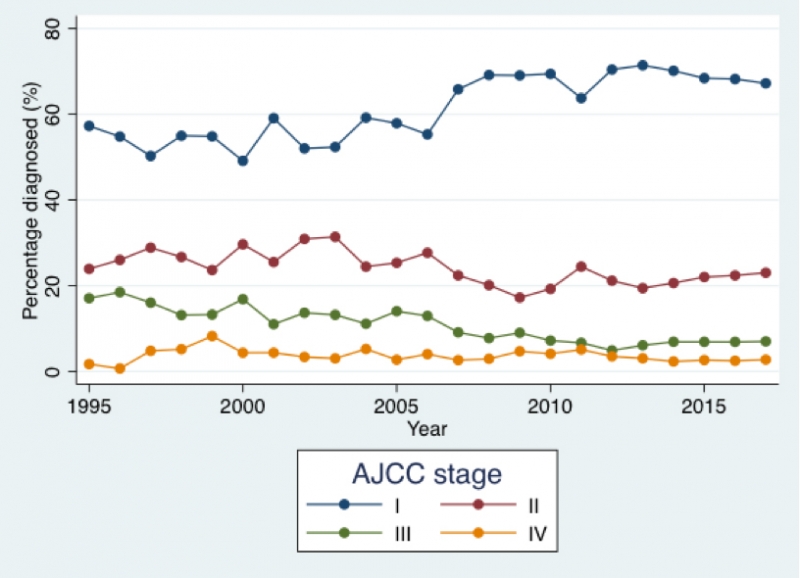
Most melanoma patients are diagnosed at stage I disease at diagnosis (reported by Cancer Research UK as having almost 100% 5-year survival). UK incidence figures generated by the Leeds group from Public Health England data show the dominance of these good prognosis thin melanomas (see graph to the right). In excess of 67% of UK patients present with stage I disease. Internationally the % is higher still.
However, the Australians have reported that more patients die from thin melanomas (<1mm thickness) than thick ones in Queensland, given the relatively higher frequency of thin tumors. The first unknown is therefore that we don’t have any means of identifying the small percentage of patients with stage I/II disease who will suffer progression of their disease. In order to allow consideration of adjuvant therapies (drugs used to reduce the risk of recurrence of the melanoma), which are established to have value but are toxic and expensive, it is an aim of MyMelanoma to develop prognostic biomarkers which can be used in the clinic to identify those patients at risk. These patients might then be better placed to understand the risks of recurrence if indeed they want to explore that (not every patient does want to) with the possible opportunity then to consider exploring the pros and cons of adjuvant therapy.
Unknown number 2: what predicts which stage III melanoma patients will benefit from immunotherapy in an adjuvant setting? Lack of a predictive biomarker for adjuvant therapies
Adjuvant targeted drug therapy and immunotherapies are offered currently in many countries if they have stage III disease. Patients considering immunotherapy would benefit from more information in order to balance the risk of possibly fatal recurrence of the cancer, with the side effects of drugs designed to help. This is especially true for adjuvant therapy (rather than treating advanced disease), as it is given to people who have a significant risk of relapse, but a proportion will be treated with toxic drugs even though they might never have developed a recurrence. The unknowns therefore relate to an inability currently to predict which stage III patients will relapse and secondly which patients are likely to respond to the drugs. The aims of MyMelanoma relating to this unknown are therefore to identify prognostic and predictive biomarkers for those adjuvant therapies: tests which predict relapse in stage III disease (prognostic biomarkers) and sceondly, failure to respond to a given drug (predictive biomarkers): both essential for patients and their medical teams in order to deliver personalized care. Studies designed to identify predictive biomarkers need to be considerably larger than those for stage IV disease, and a collaborative approach within MyMelanoma is therefore appropriate.
Unknown number 3: what do melanoma patients need to know about treatment choices and how is this information best delivered?
Health care professionals have a perspective on what patients need to know, from the questions people ask in clinic, their own family experiences and what they believe to be most important. However, patients sometimes don’t get the opportunity to express their questions and uncertainties, and people vary in what they need. An aim of MyMelanoma is therefore to collect and interpret data on what people want to know, to address those needs and then to use the most effective means of delivering answers to patients.
Unknown number 4: what lifestyle factors reduce or increase the risk of relapse of melanoma?
It has been shown that antibiotic therapies can reduce the efficacy of checkpoint blockade with parallel changes in the gut microbiome which is strong evidence identified so far that even short-term incidental changes to our bodies can affect melanoma survival. Research also suggests that diet, exercise, smoking and co-morbidities are associated with differences in the microbiome, so there is a critical need to better understand the role of lifestyle in melanoma relapse. MyMelanoma will ask how can we identify diets, patterns of exercise, and management approaches to co-morbidities which have the potential for modification to prevent relapse? What is the best advice to give a melanoma patient at diagnosis in order to reduce recurrence risk? How can this be balanced with effects on lifestyle? Every melanoma tumor is different, every person with melanoma is different, and their lifestyles are different. So very complex interactions govern who survives melanoma and how. Research to address the needs below need to involve many patients with different lifestyles, in numbers that are much, much larger than is usual for clinical trials. This is a potent justification for MyMelanoma. Guided by melanoma survivors Sean and Imogen, we identified specific key needs:-
- To understand how lifestyle affects the chance of melanoma recurring
- To develop clear information about diet, vaping, exercise and other aspects of lifestyle for patients and their families, to help them decide how to live their lives after a melanoma diagnosis
- To develop preventative interventions that patients can use to take control of their melanoma experience
Unknown number 5: how can we predict fatal or life changing toxicity, such that the toxicity can be anticipated and managed better?
There is evolving evidence that immune-related toxicity is a marker of higher response rates to immunotherapy both in therapeutic and adjuvant clinical trials. It is hypothesised that the toxicity is of prognostic significance because it is a marker of “activity” of the immune system. If that is the case, then over-zealous suppression of toxicity may be inadvisable, but predicting it would remain important if that toxicity could be fatal eg myocarditis.
There is a group of researchers working in the UK on immunotherapy toxicity led by Dr Sophie Papa and Dr Claire Palles. MyMelanoma will work with them to identify predictors of toxicity and research their treatment.
Unknown number 6: what is the nature of long-term life-changing drug toxicity for survivors?
Answering this is a MyMelanoma aim, as this is useful information of patients making decisions about therapies (particularly in an adjuvant setting, and because measures can be put in place to minimise the effect on the health of survivors.
Unknown number 7: what does having melanoma mean for patients during treatment and for survivors?
MyMelanoma aims to better establish the effects that melanoma diagnosis and treatment have on quality of life in the short and long terms so that measures can be put in place to improve those outcomes. Specifically, to appreciate what living with melanoma means for patients and their families in terms of dealing with uncertainty and in living with the impacts of treatment in the short and long term
Contact us about fundraising
To sign up to be part of MyMelanoma visit http://mymelanomastudy.org but if you have any further questions related to fund raising for us then please fill out the form. One of our team will be in touch as soon as possible.

